 |
VOLTA'S LIFE AND WORKS
Volta's localities Chronology Selected reference
 Alessandro Volta was born on February 18, 1745, in the town of Como located on a wonderful lake in north Italy, and christened on the following day, the 19th, in St. Donnino's in whose parish his family was to have its abode until 1862. His father, Filippo Volta, was of noble lineage and his mother, Donna Maddalena, came from a family of Counts, the Inzaghis. Alessandro had nine sons: four brothers and four sisters. His descent can be traced to Zanino Volta (1420) of Loveno, a little village over Menaggio on the Lake of Como, and to Martino Volta, who in the year 1518 was a merchant of Venice, trading in wool or felt on the Rialto. Alessandro Volta was born on February 18, 1745, in the town of Como located on a wonderful lake in north Italy, and christened on the following day, the 19th, in St. Donnino's in whose parish his family was to have its abode until 1862. His father, Filippo Volta, was of noble lineage and his mother, Donna Maddalena, came from a family of Counts, the Inzaghis. Alessandro had nine sons: four brothers and four sisters. His descent can be traced to Zanino Volta (1420) of Loveno, a little village over Menaggio on the Lake of Como, and to Martino Volta, who in the year 1518 was a merchant of Venice, trading in wool or felt on the Rialto.
Young Volta showed little promise for the family, for at first he appeared quite dull and slow-witted. Even at the age of four he had still not learned to talk. So back-ward was the child that his inability to form the simplest words led his family to beleave that he was actually dumb and would never be able to speak. After his fourth year, however, the glimmering of light in his mind seems to have finally caught up with itself in a quick glow of intelligence because, by age seven, he was classed among the brighter pupils of his school. This sudden improvement surprised his family and prompted his father later to say: "I had a diamond in the house but did not know it".
In the Jesuit-run schools of rhetoric he attended, he stood out among his fellow students, excelling at all times, reading any book he happened to lay his hands. He showed a special aptitude in composing easy-flowing poems and foreign languages, soon mastering Latin and French as well as his native Italian. His facility with languages soon gave Volta command with English as well as Italian, and he also learned to read German, Dutch and Spanish fluently, and Russian and old Greek. In addition to this interest in the humanities, Volta was also greatly attracted to chemistry.
Having left the Jesuits, Volta finished his schooling at the Benzi Royal Seminary in Como where he formed a friend-ship with cleric Giulio Cesare Gattoni, later to become his patron. After completing his studies, Volta became more deeply involved in his study of physics, chemistry and electricity, and having abandoned the study of low which his parents had chosen for him.
Volta began a correspondence in 1763, at age of eighteen, when he had not as yet carried out any experiments, with the eminent French physicist and electrical experimenter, the Abbé Jean-Antoine Nollet in Paris. In this exchange of letters, young Volta proposed the belief that electrical attraction followed Newtonian lows of gravitonal forces.
 In 1769, Volta sent a latin dissertation on the force of attraction of electrical fluid to Father Giovanni Battista Beccaria, professor of physics at the University of Turin and the foremost Italian experimenter in electrostatics. It was to him that Volta addressed his first publication in electrical studies. Appearing under the Latin title "De vi attractiva ignis electrici". In 1769, Volta sent a latin dissertation on the force of attraction of electrical fluid to Father Giovanni Battista Beccaria, professor of physics at the University of Turin and the foremost Italian experimenter in electrostatics. It was to him that Volta addressed his first publication in electrical studies. Appearing under the Latin title "De vi attractiva ignis electrici".
While working on the experiments carried out by Epino and other physicists on "vindice" electricity, he was struck by the test in which an electrically-charged silk ribbon is placed close to a well insulated metal plate, which led him to devise a small machine that produced a substantial number of sparks before it was necessary to recharge it.
It was in June 3, 1775, in a short letter, that Volta announced the discovery of his famous perpetual electrophorus. This he regarded as a kind of perpetual reservoir of electrical charges that worked on the novel principle of electrostatic induction or 'influence', rather than by the then well-known direct electrostatic frictional means.
This new device consisted of a cake of resin, wax, or other non-conducting substance placed between two metal plates. The resin rested on the lower plate and the upper plate had an insulated handle attached to its center which permitted it to be lefted from the resin cake.
In operation, the upper metal plate was removed and the upper surface of the resin cake was rubbed by a piece of flanel, wool or a fox tail which thus charged negative its surface. The upper plate, held by insulated handle, was placed on the resin cake; it would thus be his lower surface positive charged and negative charged the upper. By touching the top plate with a finger, the negative charge was drawn off to ground. On lifting the upper plate it would thus be only positive charged by induction. A spark could then be drawn from the plate when touched or charged a Leyden jar. When descharged, the plate could once more be placed on the resin cake and recharged and then again recharged more time without to rub the resin cake again.In 1776 Father Carlo Giuseppe Campi discovered a spring of inflammable air or hydrogen in a marshy area in the quarter of St. Colombano and asked Volta to investigate both the causes and the nature of the phenomenon. Volta could not at the time visit the spot but gave the matter some thought. As the phenomenon was to be observed in the air immediately above the swamps, he surmised that all swamps could produce inflammable air and began to study the cane thickets in Angera, on the Lake Maggiore during a fish trip (november 3, 1776).
While rowing leisurely in his boat, he noted that bubbles were rising to the surface of the lake, particularly in the shallower and more marshy locations. These were especially numerous when the bottom of the swampy parts near the shore were stirred. Volta gathered some of this gas at the lake's surface and found it to be inflammable. Up to that time, chemists had only found inflammable air in fossil coal, and rock salt deposits and were thus of the opinion that its origin was perforce to be ascribed to the mineral kingdom. Volta refuted this. In these he correctly stated that the inflammable air came from decaying animal and vegetable matter that had settled in the marches and named this air Inflammable air native of marshes. Chemists today call marsh gas Methane (CH4).
In the last of the letters he wrote to Father Campi on the matter he suggested it might be used in lamps instead of oil. He himself was the first scientist to build a perpetual lamp or Volta lamp. He applied a discharge from his new electrical invention, the electrophorus, generating a spark that to light it, or to exploded the marsh gases.
To this last device he gave the name 'electric pistole'. Volta was the precursor of exploding engine used in our cars.
 The discharge from his electric pistol gave him an idea for long distance communication. In april 18, 1777 he sent e famous letter to Professor Carlo Barletti, a teacher of physics at University of Pavia. Volta proposed an electric signal line from Como to Milan. Wooden posts were to hold an insulated iron wire over which an electric spark was to travel, then return via the canal and the lake between these two towns. Thus a Leyden jar could be discharged into the long wire at Como and detonate an electric pistol at Milan to signal a code event. Here was the germ of an idea for an electric telegraph. The discharge from his electric pistol gave him an idea for long distance communication. In april 18, 1777 he sent e famous letter to Professor Carlo Barletti, a teacher of physics at University of Pavia. Volta proposed an electric signal line from Como to Milan. Wooden posts were to hold an insulated iron wire over which an electric spark was to travel, then return via the canal and the lake between these two towns. Thus a Leyden jar could be discharged into the long wire at Como and detonate an electric pistol at Milan to signal a code event. Here was the germ of an idea for an electric telegraph.
The pistol, like the Leyden jar before it, was, however, highly popular, although Volta was surprised it should arouse such interest without anybody asking himself what possible uses it could be put to. If Volta's amazement was fully justified, it was also true that few people could understand, as he did, that it could lead to the invention of the inflammable air eudiometer.
 The properties of nitrous gas, which absorbs oxygen from the atmosphere, had led Joseph Priestley to invent the first eudiometer. Felice Fontana, Marsilio Landriani and João Hyazinthe Magellan had improved it, but Henry Cavendish had warned against many of its limitations and physicists at large avoided using it. Armand Seguin and Franz Karl Achard replaced nitrous gas with phosphorus, Claude-Louis Berthollet and Macarty with alkaline sulphides: Humphry Davy not content with this, wanted ferrous chloride to be added to nitrous gas. Volta tried using inflammable air and created an eudiometer able to record up to two thousandths of oxygen. It took him two years to perfect it. Joseph-Louis Gay-Lussac and Alexander von Humboldt compared it to other eudiometers and after close examination declared, in 1805, that Volta's eudiometer was for more accurate than all other existing ones. The properties of nitrous gas, which absorbs oxygen from the atmosphere, had led Joseph Priestley to invent the first eudiometer. Felice Fontana, Marsilio Landriani and João Hyazinthe Magellan had improved it, but Henry Cavendish had warned against many of its limitations and physicists at large avoided using it. Armand Seguin and Franz Karl Achard replaced nitrous gas with phosphorus, Claude-Louis Berthollet and Macarty with alkaline sulphides: Humphry Davy not content with this, wanted ferrous chloride to be added to nitrous gas. Volta tried using inflammable air and created an eudiometer able to record up to two thousandths of oxygen. It took him two years to perfect it. Joseph-Louis Gay-Lussac and Alexander von Humboldt compared it to other eudiometers and after close examination declared, in 1805, that Volta's eudiometer was for more accurate than all other existing ones.
In 1777 Volta travelled to Switzerland, and met Horace-Bénédict de Saussure, François-Marie Voltaire and other men of thought and distinction.
In the year 1778 he was called to occupy the chair of physics at the larger University of Pavia. At Pavia, which was 25 miles south of Milan, Volta was to teach for 27 years (1778-1805) and bring great fame to the university.
The early work of Volta at Pavia continued his strong interest in and observations of the behavior of gases. He was the first to demonstrate the nature of the expansion of gases under heat, especially at higher temperatures. His conclusions (1793) pointed strongly to a pattern of physical behavior which later became know as "Charle's law" (that all gases expand equally for equal degrees of heat). In addition, Volta diligently followed the reported vestigations of scientists on the Continent and in England particularly close of Priestley and Cavendish in their work on gases.
His command of English was of great assistance in keeping Volta informed of their published results, especially through his friend Tiberio Cavallo in London. He thus was made aware of the method developed by Cavendish of gathering pure hydrogen. Volta also observed the effect of spongy platinum on hydrogen and that, when these were brought together, spontaneous ignition of the gas would follow.
 In 1780 he visited Bologna and Florence. In 1782 he proceded to Germany, Belgium, Holland, England and France, to confer with such intellectual giants as Georg Christoph Lichtenberg, Martin Van Marum, Joseph Priestley, Antoine-Laurant Lavoisier and PierreSimon de Laplace and incidentally to enrich the cabinet at Pavia with instruments of research and demonstration. In 1780 he visited Bologna and Florence. In 1782 he proceded to Germany, Belgium, Holland, England and France, to confer with such intellectual giants as Georg Christoph Lichtenberg, Martin Van Marum, Joseph Priestley, Antoine-Laurant Lavoisier and PierreSimon de Laplace and incidentally to enrich the cabinet at Pavia with instruments of research and demonstration.
His invention of the condenser, in 1780, followed soon upon his arrival in Pavia. This valuable, higly-sensitive device draws capacitance, stores electrical energy and accumulates even the faintest traces of electricity, which would go undetected by the most sensitive electrometers. He devised it while attempting to improve the electrophorus, and produced three different versions of it. The first of the condenser consists of a white marble disc surmounted by an insulated brass disc; the second one, instead of marble, has a wooden disc with taffeta applications; the third consists of two brass discs with a slight varnish coating. Physicists prefer the marble condenser.
When in London in 1782, Volta gave a reading of his paper on the structure and the use of the condenser at the Royal Academy of Science. Twelve years later those same scientists who had been present at the Royal Academy address awarded him Copley's Gold Medal, an honour which is bestowed only an leading scientists of proven ability.
The condenser, it should be added, was to prove of invaluable use to Volta in the discovery of contact electricity.
 Thanks to the condenser and to Cavallo's electrometer, which he had perfected by replacing the elder pith pellets and methal wires with two parallele mobile straw slivers, Volta succeeded while studying the electricity produced during fermentation and evaporation, in discovering the real cause of atmospheric electricity. Thanks to the condenser and to Cavallo's electrometer, which he had perfected by replacing the elder pith pellets and methal wires with two parallele mobile straw slivers, Volta succeeded while studying the electricity produced during fermentation and evaporation, in discovering the real cause of atmospheric electricity.
Before him Franklin, Saussure, Henly and Cavallo had already investigated this phenomenon but had failed to reach any conclusion, lacking as they did the necessary instruments. He repeated the experiments in order to verify that dissolution, effervescence, evaporation and such major alterations in the mutual forces of bodies did not affect the mutual inductance of bodies.
Two years of unrelenting study ensued and, finally, he was able to prove that there is a leakage, which he gauged, when combustion, evaporation or fermentation occur, and that bodies dissolving in vapour draw electric fluid from the earth, thus producing cloud electricity.
Working on the principle of the origin of atmospheric electricity, he descovered the water found on the surface, when turning into vapour, carries electricity into the higest regions of the sky where the intense cold condenses this same vapour, releasing electricity which builds up and remains in the air until natural or artificial conductors allow it to return to the ground.
Atmospheric electricity is, as a rule, positive; when, occasionally, it happens to be negative it comes from certain clouds, which, for reasons unknown, have lost part of their electrical fluid.
The hypothesis put forward by Volta regarding the formation of hail was also noteworthy. Normally speaking, hail is a phenomenon that typically happens at the height of summer, towards midday, when the upper reaches of clouds receive direct sunlight which, along with the dry air surrounding the clouds themselves, gives rise to massive evaporation, meaning the temperature drops to such an extent as to cause raindrops to freeze.
To explain how hailstones can possibly float in the air without dropping to the ground, while becoming much heavier, Volta informs us that during thunderstorms clouds arrange themselves in different layers, having opposing charges. These layers are quite distant the one of the other, owing to the action of opposite and mutually attractive forces, proof of which is given by hideous-sounding thunderbolts that crash from one cloud to the other without falling to the ground.
The aforementioned evaporation taking place in the upper reaches of the clouds moulds these vapours into tiny snowflakes which, themselves mutually attracted and repulsed by the opposite charges holding them up, grow in volume as new raindrops add to them and freeze on their surface, and they do not fall until the weight becomes excessive or until they are no longer attracted to each other.
If hail never falls in winter and thunder is seldom heard, it is, in Volta's opinion, because reduced evaporation causes less electricity to build up, and because clouds tend to be low-lying so that terrestial conductors can more easily absorb lightning.
In the post-scriptum to a letter Volta wrote to Professor Antonmaria Vassalli-Eandi on October 24, 1795, he also broached elastic vapour. He held that the quantity of elastic vapour to be found in a vacuum or in an air-filled space, regardless of the density of the air, is constant because quantity is related purely to heat intensity, and the vapour force increases by geometrical progression while heat increases by arithmetical progression.
Volta built several prototypes of machines, meaning to describe then more fully in writing when expanding some notes he had jotted down, and these machines are still housed in the Pavia University Physics Department.
In 1801 and 1802, both John Dalton in England and Gay-Lussac in France, presumably unfamiliar with Volta's theory, although it had been reported in Brugnatelli's Chemistry Annals printed in Pavia in 1793, repeated all the experiments and applied them to all gases, be they permanent or non-permanent, obtaining the same expansion coefficient as Volta.
Gay-Lussac also gauged cubic expansion of glass and Dalton analytically expounded the theory, yet Volta remains the first scientist to have reached the aforementioned conclusions. Unfortunately, physicists at large, above all foreign ones, give Gay-Lussac and Dalton full credit for the descovery.
Volta married on September 22, 1794 at the age of 49, Donna Teresa Peregrini. He had three sons, Zanino, Flaminio and Luigi. Flaminio dead at the age of 18.
During the next five years, he wrote some of his most valuable memoirs, and secured universal fame. In 1861 it was proposed that the Regio Istituto Lombardo di Scienze e Lettere of Milan, of which Volta had been elected president in 1803, should acquire all of his available manuscripts (6000 sheets). Under the patronage of King Vittorio Emanuele II who gave 3000 Lire from his own purse, funds flowed in from villages, towns and cities of Italy and from abroad.
This drew the united efforts of many scientists and electricians who gladly parted with their original Volta letters, manuscripts and books so that they could be assembled in one place. The Brera Palace (now in the near Landriani Palace) in Milan was selected to house these scientific treasures and an index of the material was prepared.
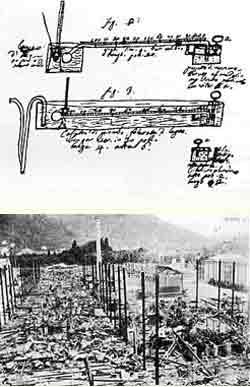 Fortunately none of this heritage, including the original letter that Volta sent to sir Joseph Banks in March 20, 1800 announcing the discovery of the pile, was sent to exposition of 1899 and it thereby escaped the disastrous fire. Fortunately none of this heritage, including the original letter that Volta sent to sir Joseph Banks in March 20, 1800 announcing the discovery of the pile, was sent to exposition of 1899 and it thereby escaped the disastrous fire.
Volta's works, collected by Vincenzo Antinori, were printed in Florence in 1816 in 5 books, with a dedication to Grant Duke Ferdinando III, while a volume of unpublished letters was issued in 1834 by the Nobili printing-house in Pesaro. Now all Volta's manuscripts and letters are collected, in 7 books his work manuscripts, in 5 books his collected letters (1918-1929); in 1966 was printed a book with the missing letters and manuscripts, and in 1974 the two index books. All this will be ready, in this year 2002 in one CD-Rom, made by Istituto Lombardo and University of Pavia.
Volta started his experimental studies on the effect of electricity in muscle contraction in 1792, after reading the recently published acounts of Galvani's experiments
 In 1786, Domenico Cotugno, Professor of Anatomy in Naples, was cutting open a live rat when the tip of the knife-blade he was using brushed against one of the animal's nerves. Although the professor suffered a shock, which he left in both arms and chest, and despite the fact that electricity was often believed to be the cause of many a natural phenomenon, it never dawned on anybody that electricity might be used to explain the incident. In 1786, Domenico Cotugno, Professor of Anatomy in Naples, was cutting open a live rat when the tip of the knife-blade he was using brushed against one of the animal's nerves. Although the professor suffered a shock, which he left in both arms and chest, and despite the fact that electricity was often believed to be the cause of many a natural phenomenon, it never dawned on anybody that electricity might be used to explain the incident.
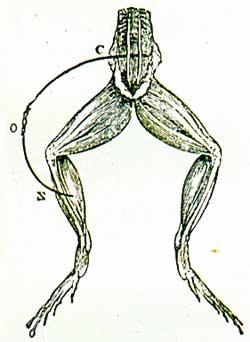
Only in 1789 did chance lead Luigi Galvani, Professor of Anatomy in Bologna, to realise the full implications of the matter. A student who was working on one of a number of skinned frogs neatly laid out next to an electrical static machine happened to touch, with the tip of his knife, the nerves of a frog's leg causing it to contract violently as if the animal were alive. Anothe student was of the opinion that the frog have moved just when the machine had emitted a spark.
This was sufficient to arouse the suspicion that the phenomenon might be due to electricity. Galvani began to carry out a series of experiments. By means of the knife he was able to transmit a spark to the frog's nerves. Galvani them tried to stimulate those same nerves directly with a spark and with the knife-blade alone.
The frog's body contracted every time, except when he used the knife by itself. Galvani then set to work on a number of different animals, both worm-blooded and cold-blooded ones, and reached the conclusion that the contractions were produced by an electric fluid particular to animal organs. He called it electric animal fluid, but physicists began to refer to it as galvanic electric fluid, as a sign of homage to its discoverer.According to Galvani, this fluid is produced in the brain by the lightest components of blood; the nerves then carry it to the different muscles, but as the nerves are but conductors, the fluid stores up in muscles which internally have a positive electric charge and externally a negative one, as is the case of the two different armatures in the Leyden jar. Every time the positive charge flows from inside the muscle of the conductor nerve to the outside of the muscle, contraction occur. Galvani published his findings in 1791, yet they were only universally known the following year.
Physicists were awed by the discovery. In a letter dated April 3, 1792, Volta defines Galvani's discoveries as wonderful. When reading his doctoral dissertation on May, 5, he expressed his admiration in even warmer tones.
Tests were carried out everywhere. Eusebio Valli confirmed the existence of the two electricities, but thought that negative elctricity is to be found inside the muscle and positive electricity outside it. Initially Volta raised no objections and wrote several papers on Galvani's electric fluid, simply stating that he believed negative electricity to be found in nerves and positive electricity in muscles, basing his theory on the observation that if the button of the Leyden jar, slightly charged, is brought close to the frog's nerves while the external armature is placed close to the muscles, this will trigger the contractions; however, fewer contractions were detected when the jar was placed in the opposite direction.
Galvani himself found nothing to object to. Yet, as René-Juste Haüy was to point out, still greater honours were to be bestowed on Volta. It was in the summer of 1792 that Volta began to entertain some doubt as to the truthfulness of Galvani and of his own hypotheses on the existence of animal electricity. Prompted to further investigation, he discovered that electricity, be it artificial or animal electricity, acts only on nerves and that electrical fluid need to flow from nerves to muscles. He also observed that electricity does not necessarily discharge from the nerve to the muscle or from the internal to the external part of the muscle; that it is sufficient to stimulate the nerve, even very briefly, for the muscle to contract; that electrical fluid can only stimulate nerves. To reach such conclusions he devised a test identical to Sulzer's wellknown test (only at a later date did he learn about the existence of Sulzer's test from Giovanni Aldini, a relative of Galvani's).
He brought a number of variations to it, creating first an acid taste an alkaline taste by changing the combination by dissimilar metals. Some times he would produce a flash of light at other time he would feel a tingling sensation or suffer abrasions or ulcers as a result of his experiments. He even tried, but to no avail, to stimulate his sense of smell. All these experiments, he said, do not prove conclusively that animal electricity exists, since the organs remain passive, whereas metals are always active, be they identical or dissimilar, when carefully applied to wet portions of skin, and ensure the flow of electricity through the body, provided contact is established. He also wrote that he had obtained the passage of electrical fluid to water-sodden materials or to water itself. He concluded: It is therefore the metals that apparently cause the electricity to flow, which means that they are not simply deferents, as at other times, but proper movers and exciters of electricity, a discovery of major importance. It therefore remains to be assessed whether the contractions and muscular spasms observed in the animals used by Galvani can be attributed to a form of electricity peculiar to the organs. Although initially convinced of this, I am now strongly inclined to disbelieve it.
His doubts were corroborated by the fact that animal spasms never occured, or hardly ever occurred, if not when contact was made with some metal, or better, two dissimilar metals. Little remained of the animal electricity described by Galvani, if not the extreme sensitivity of nerves (responsible for physical sensation and movement), to electrical stimuli produced by some external source.
 Late in 1792 the Leipzig Scientific Journal carried an article on animal electricity. In this article Volta briefly described the phenomena he had observed, then went on to state that such phenomena are all too easily ascribed to artificial or external electricity produced by mutual contact between metals and coal. Late in 1792 the Leipzig Scientific Journal carried an article on animal electricity. In this article Volta briefly described the phenomena he had observed, then went on to state that such phenomena are all too easily ascribed to artificial or external electricity produced by mutual contact between metals and coal.
In 1794 in a letter to Vassalli, he brought new evidence, which he thought to be so conclusive that he decided to call this form of electricity not animal electricity, but metallic electricity. One of the scientists who most convincingly argued against Galvani's theory was Christoph Heinrich Pfaff, professor at Kiel University, but Libes is sadly wrong when he argues Pfaff was the first person to refute Galvani. Volta had been doing so since the summer of 1792 and it was only in 1793 in Stuttgart that Pfaff published his first dissertation on animal electricity, presumably after learning about Volta's doubts from the Leipzig journal.
Volta had brilliantly demonstrated that apart from metals, bodies containing a certain amount of metals and some kinds of caol, no other conductor leaves an electric taste on the tongue, or produces a flash of light or causes any spasms in the legs of frogs, even if they have just been killed. If electricity was peculiar to these animals, why was it not produced by other good conductors, apart from metals and coal, once the circuit was formed, and why were no spasms detected? All the magic, and do allow me the term, said Volta, belongs to the class of metals and to some vegetable and animal coals having some affinity with these metals It is obvious that one of the main prerequisites lies in the dissimilarity of the metal conductors that, together with animal matter, form the circuit.
Aldini, Valli and other advocates of the animal theory pointed out that one metal is all that is needed to stimulate animal electricity and to induce the spasms in frogs, as it acts as an ideal conductor to favour the flow from nerves to muscles. Volta disproved this, thus adding to his renown. Tempera colours, rust, heat and other similar causes make different parts of the same knife so different the one from the other that it may well be constituted by dissimilar metals, which explains why a knife alone may be sufficient. When a knife which had unsuccessfully been used to stimulate some reaction in frogs was coated with tempera, the frogs contracted as if alife. Which metal is purer than silver when it is polished? Even the purest quicksilver, just because its external surface oxidizes in air while its core undergoes no change, can cause the contractions. It is one of the primary electrical laws that bodies whose surface changes or are rubbed with different substances, have different electrical charges.
In the absence of metals and coal it is possible to induce the bodies of frogs to contract simply by placing two different parts of the animal's bodies close to one another, provided they are sufficiently dissimilar or covered in some substance that acts as a conductor. If Volta chose the name metallic, it is not because the newly-discovered form of electricity could be produced only by metals, but because metals had proved to be the ideal movers. For this reason he called them first-class movers and conductors, so that they may not be confused with other bodies having, though to a lesser degree, the same properties, and Volta concluded that the fluid detected is not animal fluid, but common electricity.
Volta also discovered that not all metals behaved with the same degree of effectiveness and that some were more capable of stimulation than others. He drew up a list of these metals and arranged them in the order of their effectiveness; this list was published in 1794. In it he placed zinc first, then tin, lead, iron, copper, platinum, gold, silver, graphite, charcoal. Volta was then able to state one of the most important laws in the whole science of electricity; if two of the elements in the above list were joined they would produce a more effective result the farther apart they were in the list. This meant that zinc and charcoal could produce stronger results than the combination of any other two materials. This same list later became known as the electrochemical series, a very important tool for the practicing electrical designer and engineer as well as every practicing chemist.
The conditio sine qua non is that three different conductors be present in the circuit, or two dissimilar metals that, touching at one extremity, communicate at the other extremity with one or more moist conductors, which will give a total of three moist conductors all dissimilar from each other. Volta warned that of all the possible methods of producing electricity, this is the least effective.
Between 1794 and 1798 he dictated a number of papers in which he expounded his theories and adduced more and more evidence, such as in the letters he wrote to Vassalli and Aldini and in the letter he wrote in 1796 to Professor Gren in Halla, which was published in the German newspapers. He held a number of public demonstrations in his laboratory in Pavia and private ones, surrounded by Italian and foreign friends alike, in his private apartment. Humboldt wrote about them in his letter to Professor Johann Friedrich Blumenbach on August 26, 1795, and this was also given to the press. All this is proof that Volta was always ready to share his ideas with other distinguished physicians, although nobody had any inkling of the work which was to lead to the invention of the cell.
It was only in august 1796 that Volta chanced upon the happy idea of using the electrometer and the condenser to study the unusual motive force of electrical fluid. This was to be an important step towards the invention of the cell. Using this equipment, and occasionally replacing the condenser with Nicholson's duplicator, he descovered that when silver is placed next to tin electrical fluid flows from the first to the second metal. If moist conductors are present, and the circuit is complete, the flow of electricity is constant and always in the same direction. If the circuit is incomplete and the metals are insulated, the fluid builds up in the tin rather than the silver, and the former has a positive electric charge while the latter has a negative one. In a circuit comprising first and second-class conductors with couples of dissimilar metals, each of which moving the electric flow in one direction, those forces made to move in the opposite direction are destroyed while the forces are mutually strengthened if they maintain their pristine direction.
Electricity is infinitely tenuous, but it condenses enough to produce a spark. The more metals differ, the more vigorously they will produce electricity. By means of the duplicator, traces of electricity can be obteined even from mere contact between dissimilar conductors. Volta repeated the experiments already effected with silver and tin, but using other metals, conductors and motive objects. Based on the principle of their reciprocal quality, Volta compiled a series comprising a score of combinations, and one can see, as if in a mirror, when frogs would be affected by convulsive movements and when they would not. He obteined traces of negative electricity from a metal blade, a wooden ruler and other matters that were well insulated and briefly exposed to sunlight or fire, or simply put in a worm place. The traces were stronger if the objects were bung by a silk cord and made to spin in the air, like a sling, for two or three minutes. Positive electricity manifested itself if the same objects were put in a colder and damper place than usual.
He obtained electricity merely from the contact of insulated metal with a different kind of metal, whether insulated or not, and in 1797 he got immediate electricity traces from the same metals even without making use of the condensers. The wider the surfaces in contact the more evident the traces, while if the two metals only touched each other at two points, as would happen with two spheres, then the electricity moved was so negligible that it was hardly detected by using the duplicator, let alone the condenser.
After such imposing facts, the road to the Voltaic Pile was truely brief. It was known that the wider the surfaces employed, the more numerous the contact points were and, therefore, the stronger the traces of electricity. It should have been easy to imagine a device for putting together many of those metal surfaces, in the form of couples, interposing a conductor between them and constructing a Pile by adding their energy together.
   It was undoubtedly an easy step if we judge it from today's position, but after learning of the effect of lenses in spectacles, what could have been easier than inventing the telescope? No matter, three centuries elapsed between one invention and the other, while it took Volta only three years. It was undoubtedly an easy step if we judge it from today's position, but after learning of the effect of lenses in spectacles, what could have been easier than inventing the telescope? No matter, three centuries elapsed between one invention and the other, while it took Volta only three years.  He was engaged in fighting Galvani's hypothesis and reconfirming his own theory , and he was not willing, or able, to fcus his mind on a higher aim. The results he produced were sufficient for Volta to establish his theory, but not content he went on inquiring into the method of greatly multiplying the metal couples and constructing a small machine which could increase the strength or electricity charge to a high degree. The forces by which the silver could send the electricity fluid upwards and downwards into the zinc would balance out, although he would not have solved the problem even by using three or more dissimilar metals, since in every series of dissimilar metals the electric tension at both ends is exactly the same as that obtainable from the contact between the first and the last metal, and therefore all the intermediate metals are useless. He tourned these thoughts over in his mind during 1797 and 1798 and for the most part of 1799, then he decided to interpose a moist layer, or second-class conductor, between the metal couples so that they could communicate with one another. The action of moist conductors with metals is usually very weak and is either not equal to the mutual action between metals themselves, or it can change the action very little, whether decreasing or increasing it. He was engaged in fighting Galvani's hypothesis and reconfirming his own theory , and he was not willing, or able, to fcus his mind on a higher aim. The results he produced were sufficient for Volta to establish his theory, but not content he went on inquiring into the method of greatly multiplying the metal couples and constructing a small machine which could increase the strength or electricity charge to a high degree. The forces by which the silver could send the electricity fluid upwards and downwards into the zinc would balance out, although he would not have solved the problem even by using three or more dissimilar metals, since in every series of dissimilar metals the electric tension at both ends is exactly the same as that obtainable from the contact between the first and the last metal, and therefore all the intermediate metals are useless. He tourned these thoughts over in his mind during 1797 and 1798 and for the most part of 1799, then he decided to interpose a moist layer, or second-class conductor, between the metal couples so that they could communicate with one another. The action of moist conductors with metals is usually very weak and is either not equal to the mutual action between metals themselves, or it can change the action very little, whether decreasing or increasing it.
He therefore constructed a small machine using a series of first and second-class conductors, such as silver, zinc, and a moist layer, and then he repeated the same sequence, as many times as he wanted. In this way he found what he had thought to find. The electricity tension increases exactly as much as the number of couples in the series, and if the tension of one couple is a sixtieth of a degree on the electrometer, it gains double, triple, quadruple strength when two, three or four couples are used.
This is the great step I took, Volta said, at about the end of 1799, a step that soon led me to the construction of the new shock-giving apparatus
. Which was the cause of so much amazement for all the physicists, and gave me high satisfaction followed by amazement not long after the aforementioned discovery that held such a promise of success'.
What first surprised him was the electric shock, which he felt in his hands and arms that were acting as an arc at the two ends of the small machine. Around January 1800 he made the first of these small machines in Como, and on March 20, 1800 he gave notice of it to Joseph Banks, President of the Royal Society of London. Here a little beginning part of the letter what Volta wrote:
After a long silence, for which i shall offer no apology, I have the pleasure of communicating to you, an through you to the Royal Society, some striking results I have obtained in pursuing my experiments on electricity excited by the mere mutual contacts of different kinds of metal, and even by that of other conductors, also different from each other, either liquid or containing some liquid, to which they are properly indebted for their conducting power. The principal of these results, which comprehends nearly all the rest, is the construction of an apparatus having a resemblance in its effects (that is to say, in the shock it is capable of making the arms, &c. experience) to the Leyden flask, or, rather, to an electric battery weakly charged acting incessantly, which should charge itself after each explosion; and, in a word, which should have an inexhaustible charge, a perpetual action or impulse on the electric fluid; but which differs from it essentially both by this continual action, which is peculiar to it, and because, instead of consisting, like a common electric jars and batteries, of one or more insulating plates or thin strata of those bodies which are alone though to be electric, armed with conductors , or bodies called non-electric, this new apparatus is formed merely of several of the latter bodies, chosen from among those which are the best conductors, and therefore the most remote, as has hitherto been believed, from electric nature.
The apparatus to which I allude, and which will, no doubt, astonish you, is only the assemblage of a number of good conductors of different kinds arranged in a certain manner. 30, 40, 60, or more pieces of copper, or rather silver, applied each to a piece of tin, or zinc, which is must better, and as many strata of water, or any other liquid which may be a better conductor, such as salt water, lye, &c. or pieces of pasteboard, skin &c. well soaked in these liquids; such strata interposed between every pair or combination of two different metals in an alternate series, and always in the same order of these three kinds of conductors, are all that is necessary for constituting my new instrument, which, as I have said, imitates the effects of the Leyden flask, or of electric batteries, by communicating the same shock as these do; but which, indeed, is far inferior to activity of these batteries when highly charged, either in regard to the force and noise explosions, the spark, the distance at which the discharge may be effected, &c. as it equals only the effects of a battery very weakly charged, though of immense capacity; in other respects, however, it far surpasses the virtue and power of these batteries, as it has no need, like these, of being previously charged by means of foreign electricity, and as it is capable of giving a shock every time it is properly touched, however often it may be.
To this apparatus, much more similar at bottom, as I shall show, and even such as I have constructed it, in its form to the natural electric organ of the torpedo or electric eel, &c. than to the Leyden flask and electric batteries, I would with to give the name of the artificial electric organ: and, indeed, is it not, like it, composed entirely of conducting bodies? Is it not also active of itself without any previous charge, without the aid of any electricity excited by any of the means hitherto known? Does it not act incessantly, and without intermission? And, in the last place, is it not capable of giving every moment shocks of greater or less strength, according to circumstances shocks which are renewed by each new touch, and which, when thus repeated or continuied for a certain time, produce the same torpor in the limbs as is occasioned by the torpedo, &c.?
I shall now give a more particular description of this apparatus and of others analogous to it, as well as of the most remarkable experiments made with them.
I provide a few dozens of small round plates or disks of copper, brass, or rather silver, an inch in diameter
..
The French, owning to its shape, and in order to honour Galvani, called it La Pile de Galvani, while others, more rightly, named it La Pile de Volta. In the end, the name of Pile, being simpler, prevailed.
Volta thought about how to vary it by using glass cups in which, in the same fashion as in the Pile, there is the same sequence of metals and moist conductors. This variation is known as the Cup Crown apparatus.
This revolutionary contribution, one of the most brilliant gifts of the human mind, was immediately recognized for its true importance. Experimenters everywhere were now afforded a source of constant-flow electricity. They found in these new devices a means of drawing electric current for hours instead of the erratic spark that came from the electrostatic generators or Leyden jars in use for a century.
With this new instrument William Nicholson and Antony Carlisle in England decomposed water into its elements and determined the true volumetric ratio of oxygen and hydrogen. Sir Humphrey Davy, using a large voltaic pile, discovered potassium and sodium. He also drew an electric current from a 500-plate voltaic battery and caused two charcoal electrodes to burn with sun-like brilliance; in this way began electric illumination. With constant flow electricity the electro-magnet was formed by Jean-François-Dominique Arago and by Davy. Thus the last century began punctually with the significant forward move that brought electricity from a plaything of the curious to a most important tool in the hands of mankind.
Succeeding generations of electricians, who best understood the magnitude of Voltas contribution, saw fit to measure electromotive force by the term volt as proposed by the International Electrical Congress meeting in Paris in 1881. In his eulogy of his colleague Volta, Arago wrote of the electric pile as the most marvellous instrument created by the mind of man, not excluding even the telescope or the steam engine.
 At the invitation of Napoleon I, Volta went to Paris in 1801 and performed experiments and a series of three lectures on November 7, 12 and 22, before the Institut de France. That these lectures were considered highly important was indicated by the presence of the diplomatic corps, of the First Consul of France, Napoleon Bonaparte himself and a great number of french scientists. At the invitation of Napoleon I, Volta went to Paris in 1801 and performed experiments and a series of three lectures on November 7, 12 and 22, before the Institut de France. That these lectures were considered highly important was indicated by the presence of the diplomatic corps, of the First Consul of France, Napoleon Bonaparte himself and a great number of french scientists.
Writing later to his old brother, Volta said, "Bonaparte was in good humor, at ease and gracious, and the conversation lasted more than an hour and a half. I, myself, joking aside, am amazed how my old and new discoveries of the so-called galvanism, which show them to be only pure and simple electricity caused by the contact of metals, could have produced so much excitement. Objectively regarded, I find them also of some importance, they certainly throw new light on the theory of electricity. They open a new field for chemical research and also offer applications to medicine. For a year or more the journals of Germany, France and England have been full of discussions about them. Here in Paris this is the current excitement where, as in other things, there is added the excitement of fashion."
As for Bonaparte, he was delighted with the exhibition. At the end of Volta's first lecture, that of November 7, Bonaparte, turning to his physician Nicolas Corvisart, exclaimed, Here, my good doctor, we have the image of the life itself! The pile represents the column of vertebrae, the liver is the negative pole, the kidneys, the positive pole Five days later Napoleon was again present along with his minister Jean-Antoine Chaptal. Now Bonaparte himself helped with the experiments, drawing sparks from the pile, melting a steel wire, discharging an electric pistol and decomposing water into its elements.
So impressive was Volta's presentation that shortly thereafter the Italian scientist was made one of the eight foreign members of the Institut de France and was created a Knight Commander of the Legion of Honor. Two additional awards were them made by the Institut to their honored guest for, counter to its usual custom, it voted by acclamation to present its gold medal to Volta in recognition of the very important scientific discoveries which had been presented before the Institut. In addition, a purse of 6000 francs was awarded to him for the expenses of the journey and as further encouragement to continue his scientific research.
Moreover, some years later, Napoleon, still not satisfied with the honors and rewards he had already conferred on volta, raised him to the position of Count and had him made a Senator of the Realm of Lombardy.
Not only as First Consul but also later Emperor, Napoleon added to the many honors conferred on Volta by his personal interest in the man from Pavia. When Bonaparte visited Italy in 1804, he stopped for a short time to receive a delegation of learned members of the Institute of Italy. The Emperor looked for Volta in the front ranks and, not seeing him there, made quick inquiries as to where he was, showing his disappointment in strong terms. On learning that Volta was considering retirement, Napoleon would not permit it and ordered that even though Volta gave only one lecture a year, that would be sufficient. If, said Bonaparte, Volta's name were not among the list of professor of the University of Pavia, it would be as if struck to the heart; for the rest, a good general ought to die on the field of honor.
Again, on Napoleon's third visit to Italy, he stopped at the University of Pavia, called on Volta and, before his class, placed his hand on the professor's shoulder and exclaimed, "Bravo, Volta! Bravo! You are a worthy instructor of youth!"
So impressed was Bonaparte by the novelty and promise of Volta's demonstrations that shortly after the session of the Paris Accademy he made the following announcement: "As encouragement to further experimentation and discovery, I wish give the sum of 60.000 francs to the one who will give to electricity and galvanism the advances in this field equivalent to those already given to these sciences by Franklin and Volta. My special aim is to encourage and to fix the attention of physicists on this branch of physics which is, in my opinion, the road to great discoveries." This proposal, made on June 15, 1802, was enthusiastically received by the scientists of France.
Realizing Volta the generation of electricity by the contact of dissimilar metals, that Volta's contibutions represented only the beginning of discovery in the field of science, and of electricity in particular, a special commission was created to engage in these studies. This commission included the most brilliant men of science then living in France. Laplace, eminent astronomer-mathematician; Coulomb, military engineer, physicist and electrical experimenter; Fourcroy, France's foremost chemist; Vauquelin, chemist, discoverer of chromium and beryllium compounds; Brisson, physicist, chemist and experimenter in magnetism; and Biot, mathematician and physicist who was made chairman of this important commission. Their report was issued at the end of the year and Volta was again accorded the recognition which he so well deserved. Three members of the commission had already served on a seven-man board which had, in 1797, made a similar study and had rendered its report on galvanism. These reports did much to reduce the heat of controversy and re-focus attention on the important possibilities that the voltaic pile presented.
Volta, in his hour of triumph, continued to refer to a whole family of new electrical phenomena as 'galvanism' and was always ready to acknowledge his debt to Galvani with deep respect and admiration.
In the year 1819, he established himself in Como, to spend the rest of his life in the bosom of his family. In July, 1808 in vain had the Russian Emperor called him to Petersburg with rich offers.
Volta died, in his house in Como, when he was 82 years old, peacefully, surrounded by his faithful wife and his two sons. On March 5, 1827 he closed his days spent for the scientific research, for the study of the phenomena free in nature, and partly devoted to political engagements for his home-town. A slight persistent fever befell him and being already tired and worn-out he died in few days.
In Paris, on the same day, Volta's old colleague Pierre-Simon de Laplace, also died.
Volta's biographers describe him as tall, large-brow and noble of mien, of regular features; and, according to Arago, of country manners contracted in his youth. So far as his manners are concerned, Arago was probably wrong, for the evidence discloses a philosopher singularly gracious, fair-minded, and companionable.
Volta's localities Chronology Selected reference
|
 |



 In 1769, Volta sent a latin dissertation on the force of attraction of electrical fluid to Father Giovanni Battista Beccaria, professor of physics at the University of Turin and the foremost Italian experimenter in electrostatics. It was to him that Volta addressed his first publication in electrical studies. Appearing under the Latin title "De vi attractiva ignis electrici".
In 1769, Volta sent a latin dissertation on the force of attraction of electrical fluid to Father Giovanni Battista Beccaria, professor of physics at the University of Turin and the foremost Italian experimenter in electrostatics. It was to him that Volta addressed his first publication in electrical studies. Appearing under the Latin title "De vi attractiva ignis electrici". The properties of nitrous gas, which absorbs oxygen from the atmosphere, had led Joseph Priestley to invent the first eudiometer. Felice Fontana, Marsilio Landriani and João Hyazinthe Magellan had improved it, but Henry Cavendish had warned against many of its limitations and physicists at large avoided using it. Armand Seguin and Franz Karl Achard replaced nitrous gas with phosphorus, Claude-Louis Berthollet and Macarty with alkaline sulphides: Humphry Davy not content with this, wanted ferrous chloride to be added to nitrous gas. Volta tried using inflammable air and created an eudiometer able to record up to two thousandths of oxygen. It took him two years to perfect it. Joseph-Louis Gay-Lussac and Alexander von Humboldt compared it to other eudiometers and after close examination declared, in 1805, that Volta's eudiometer was for more accurate than all other existing ones.
The properties of nitrous gas, which absorbs oxygen from the atmosphere, had led Joseph Priestley to invent the first eudiometer. Felice Fontana, Marsilio Landriani and João Hyazinthe Magellan had improved it, but Henry Cavendish had warned against many of its limitations and physicists at large avoided using it. Armand Seguin and Franz Karl Achard replaced nitrous gas with phosphorus, Claude-Louis Berthollet and Macarty with alkaline sulphides: Humphry Davy not content with this, wanted ferrous chloride to be added to nitrous gas. Volta tried using inflammable air and created an eudiometer able to record up to two thousandths of oxygen. It took him two years to perfect it. Joseph-Louis Gay-Lussac and Alexander von Humboldt compared it to other eudiometers and after close examination declared, in 1805, that Volta's eudiometer was for more accurate than all other existing ones. In 1780 he visited Bologna and Florence. In 1782 he proceded to Germany, Belgium, Holland, England and France, to confer with such intellectual giants as Georg Christoph Lichtenberg, Martin Van Marum, Joseph Priestley, Antoine-Laurant Lavoisier and PierreSimon de Laplace and incidentally to enrich the cabinet at Pavia with instruments of research and demonstration.
In 1780 he visited Bologna and Florence. In 1782 he proceded to Germany, Belgium, Holland, England and France, to confer with such intellectual giants as Georg Christoph Lichtenberg, Martin Van Marum, Joseph Priestley, Antoine-Laurant Lavoisier and PierreSimon de Laplace and incidentally to enrich the cabinet at Pavia with instruments of research and demonstration. Thanks to the condenser and to Cavallo's electrometer, which he had perfected by replacing the elder pith pellets and methal wires with two parallele mobile straw slivers, Volta succeeded while studying the electricity produced during fermentation and evaporation, in discovering the real cause of atmospheric electricity.
Thanks to the condenser and to Cavallo's electrometer, which he had perfected by replacing the elder pith pellets and methal wires with two parallele mobile straw slivers, Volta succeeded while studying the electricity produced during fermentation and evaporation, in discovering the real cause of atmospheric electricity.